SPE columnsSPE columns, CHROMABOND PFAS, 6 mL/300 mg
RM0.00
Special phase for SPE analysis. For enrichment of PFAS from water, textiles and sediments (contaminated soils). Polymeric combination phase with weak anion exchanger, highly porous, spherical particles.
Package unit 30 Piece(s)
- Description
- Additional information
Description
- Technical Data
- Learn more
- Downloads
| Hazardous material | No |
| Method | Solid phase extraction (SPE) |
| Brand | CHROMABOND |
| Phase | PFAS |
| Mode | Reversed phase (RP) |
| Base material | Polymer-based |
| Surface chemistry | Special combination phase with weak anion exchanger |
| Column type | SPE column |
| Hardware | Polypropylene (PP) columns with polyethylene (PE) filter elements |
| Column shape | Open tubular syringe-shaped column with Luer tip outlet |
| Column volume | 6 mL |
| Filling quantity | 300 mg |
| Column volume/adsorbent weight | 6 mL/300 mg |
| Recommended application(s) | Per- and polyfluoroalkyl substances (PFAS) from water, PFAS from sediments (contaminated soils), PFAS from textiles |
| Particle type | Fully porous particles(FPP) |
| Particle size | Not available or not applicable |
| Particle shape | Spherical |
| Pore size | Not available or proprietary |
| Specific surface according to BET | Not available or proprietary |
| pH stability | 1.0–14.0 |
| Filter element material | Polyethylene (PE) |
| Storage temperature | RT |

What you need to know about PFAS and PFAS analysis
What are PFAS?
More than 4730 compounds(1) belong to the group of PFAS (which stands for per- and polyfluoroalkyl substances) that have been produced since the 1940s. Since these compounds do not originate from nature, the global pollution is the result of human activity. All PFAS are of anthropogenic origin. PFAS are “forever chemicals”, chemicals that are very persistent in the environment and in the human body.
General structure of per- and polyfluoroalkyl substances (PFAS)
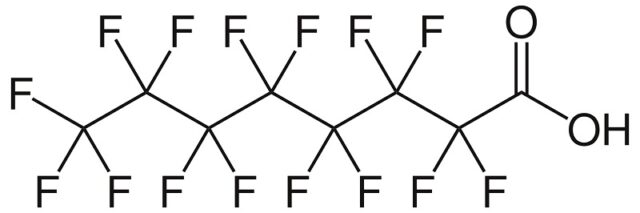
PFAS are organic compounds with a carbon chain in which hydrogen is substituted by fluorine. The carbon-fluorine bond is very strong which makes them “virtual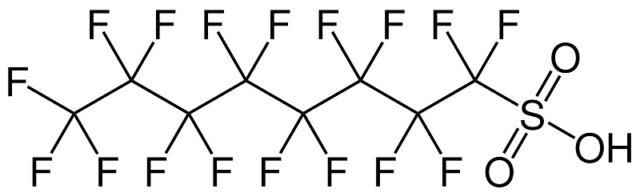 ly indest ructable“. The molecular structure of the PFAS provides them with non-sticky and tensid-like characteristics (because of their hydrophobic, lipophobic chain + hydrophilic head).
ly indest ructable“. The molecular structure of the PFAS provides them with non-sticky and tensid-like characteristics (because of their hydrophobic, lipophobic chain + hydrophilic head).
There are polymers and non-polymers. Typical polymers are fluoropolymers, side-chain flurorinated polymers and perfluoropolyethers. Typical non-polymers are perfluoroalkyl acids (PFFFAs), perfluroalkane sulfonyl fluorides (PASF), perfluroalkyl iodides (PFAIs) and per- and polyfluroalkyl ether (PFPEs) based derivatives(2).
Perfluorooctanoic acid (PFOA) and perfluorooctanesulfonic acid (PFOS) have been the most produced and studied of these chemicals.
To cut a long story short: there are many different substance groups that need to be analyzed!

How do PFAS find their way into the environment and the human body?
Per- and polyfluoroalkyl substances (PFAS) have been manufactured for more than 80 years, but health effects were neglected for a long time. In September 2020, the European Food Safety Authority (EFSA) published a new health risk assessment related to the presence of PFAS in food(3). This is the first EFSA expert opinion in which, in addition to PFOA and PFOS, other PFAS were also included in the exposure assessment and health risk assessment.
PFAS are emitted into the environment by different pathways. For example, exhaust air from industrial sources can contain PFAS and thus are dispersed into nearby ground and water bodies. Rain and snow, for example, can eventually carry them from the air into the soil and surface waters. Particle accumulation can even cause them to travel long distances through the air. PFAS are therefore also found far from industrial production sites and human living areas, such as in sediments from the Bering Sea to the Arctic(4/5). Through volatilization from products (evaporation from carpets or home textiles treated with soil-repellent agents) or from waterproofing sprays, indoor air can also be contaminated.
Soils can also be directly contaminated, for example by firefighting foams. With the uptake of PFAS from contaminated soils and waters in vegetation and their accumulation in fish, these substances enter the human food chain. Consequently, humans absorb PFASs from the environment through food, water or air.
These “forever chemicals” also find their way into wastewater treatment facilities from household sources. They then enter surface waters via treated wastewater or remain in sewage sludge. The sewage sludge, in turn, can be used as fertilizer in agriculture, and then over time these chemicals eventually leach into the groundwater. Once there, some of the precursor compounds are transformed into the persistent PFAS.

The new special phase – CHROMABOND PFAS
Over the years many different PFAS were developed. Now, they are found in the environment (water, food, soil, animals and humans) and their problematic health effects come into play.
The challenge is that current analytical methods are limited.
To tackle this challenge, we developed a special phase for the enrichment of a broad range of PFAS which provides good reproducibility and high recovery rates.
This is possible due to the different interactions the sorbent combination offers. These interactions are recommended by DIN 38407-42, EPA 537.1 and 533 guidelines.
Our CHROMABOND PFAS is a polymer-based combination phase which contains a weak anion exchange functionality. The combination of different SPE phases makes it possible to use various interactions (dipole-dipole, ionic, hydrophobic, H-bond).
CHROMABOND PFAS provides several advantages
- Solution for various PFAS substance classes
- > 28 PFAS can be enriched
- Sorbent retention mechanisms according to DIN 38407-42, EPA 537.1 and 533 guidelines
- High capacity
- High recovery rates
Additional information
| Selling unit | 30 columns, 250 columns |
|---|

